
Understanding Free Radicals
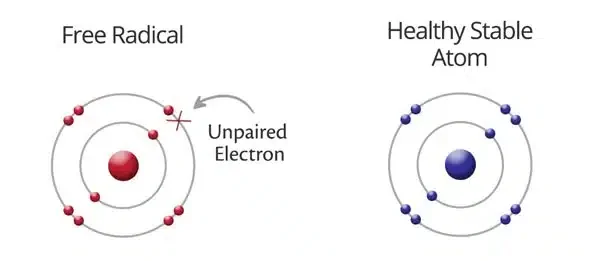
Free radicals are unstable molecules with an unpaired electron in their outer shell, making them highly reactive and potentially damaging to cells and tissues. They can exist in three states: positively charged, negatively charged, or neutral. The presence of the unpaired electron is what makes them so reactive. These radicals can occur naturally, for instance, through UV radiation splitting chlorine molecules or through high temperatures causing homolytic fission. Additionally, chemical reactions, particularly redox reactions, are another source of free radicals.
Insights on Free Radicals in Physiology
The Unstable Nature of Free Radicals
- A free radical, born from molecular cleavage, possesses a unique characteristic: an unpaired electron. This odd electron makes it highly reactive and unstable.
- Driven by this instability, free radicals actively interact with neighboring molecules, setting off a chain reaction of interactions.
- Their core behavior revolves around gaining or losing an electron, transferring their reactive nature to other molecules and triggering damaging chain reactions.
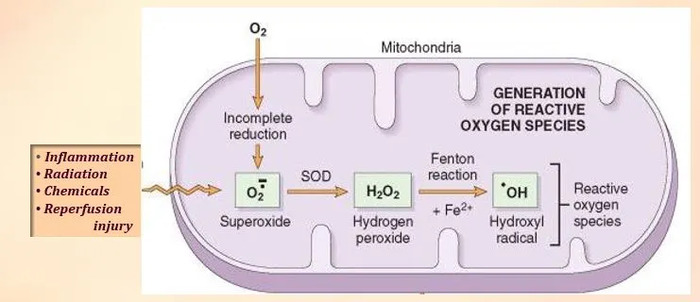
Mitochondria: A Source of Reactive Oxygen Species (ROS)
- Within the intricate world of cells, particularly in mitochondria, reactive oxygen species (ROS) – a subgroup of free radicals containing oxygen – originate.
- These ROS form during various physiological processes like respiration, stress responses, and inflammation.
- For example, during cellular respiration, an oxygen molecule gains an electron, transforming into a superoxide anion, a prime example of a free radical.
- This complex interplay highlights the crucial role of free radicals in shaping various biological functions, linking molecular instability to diverse physiological events.
Oxygen’s Double-Edged Sword: ROS and RNS
- ROS, a subset of free radicals, includes prominent examples like hydroxyl radicals, superoxide anions, and hydrogen peroxide (H2O2).
- Mitochondria, heavily involved in energy production through oxidative phosphorylation, are major contributors to superoxide anion production, which can further form hydrogen peroxide.
- Cells with high metabolic rates tend to produce more ROS.
- In contrast, reactive nitrogen species (RNS) are another type of free radical that doesn’t contain oxygen. Nitric oxide (NO) is the most prevalent RNS, synthesized during the breakdown of L-arginine.
How Free Radicals are Formed
| – Radicals, characterized by unpaired valence electrons, form as a consequence of diverse factors such as chemical reactions, radiation exposure, or environmental influences. For example, the hydroxyl radical (HO•) originates from a water molecule with a missing hydrogen atom, resulting in one oxygen bond dangling: H2O (Loss of H )⟶ H−O• – The carbene molecule (CH2) exemplifies radical formation with two dangling bonds, a process that involves the removal of specific groups: Removal of Groups⟶:CH2 – In a parallel manner, the superoxide anion (•O−2) is generated by adding an extra electron to an oxygen molecule (O2), introducing a dangling bond: O2+e−⟶•O−2 – These radicals play a crucial role in shaping chemical reactions and biological processes, contributing significantly to molecular dynamics. The presented formulas provide a glimpse into the stepwise mechanisms underlying the formation of these important radical species. |
ROS: Balancing Repair and Dysfunction
| Mitochondria: Source of Free Radicals (ROS) Cellular respiration in mitochondria can convert oxygen (O₂) into superoxide anion (O₂⁻), a free radical, as oxygen interacts with electrons. The Double-Edged Sword of ROS: Adequate ROS levels: Play a role in cellular repair, controlled cell death, and immune response. High ROS levels: Can trigger inflammation, metabolic dysfunction, and DNA damage. |
key points regarding free radicals
Beneficial Role of Free Radicals:
- Free radicals, when maintained at a controlled level, play a crucial role as defenders of our cells.
- Analogous to “indispensable friends” or “soldiers,” they contribute to the immune system’s defense against attacking microorganisms.
Physiological Production of Free Radicals:
- The immune system activates the production of free radicals in response to inflammatory processes.
- This activation is a natural and physiological response, targeting microorganisms for elimination.
Functions at the Cellular Level:
- Free radicals serve various essential functions at the cellular level.
- They participate in intracellular communication and contribute to vital biological processes, including energy production and fatty acid metabolism.
Why Free Radicals May Pose Harm
Despite their crucial functions in supporting the body’s processes, free radicals can become a threat to our health due to their inherent characteristics.
Unstable and Reactive Nature
The danger arises from the inherent instability and high reactivity of free radicals, characterized by the presence of a single, unpaired electron in their last orbital. This “unpaired” electron drives the molecules to easily bond with nearby molecules in an attempt to regain stability.
Chain Reactions
Free radicals, driven by the quest for stability, initiate chain reactions by extracting electrons from neighboring molecules. This electron “theft” transforms the molecules they interact with into free radicals, perpetuating a cascade effect. These chain reactions have the potential to impact various cellular components, including lipids, carbohydrates, proteins, and genetic material such as DNA and RNA.
Oxidative Stress
When the production of free radicals overwhelms the body’s antioxidant defenses, an uncontrolled process known as oxidative stress ensues. In this state, electrons are continually stolen, causing cumulative damage at the molecular level. This damage extends to cells and tissues, representing a significant threat to overall health.
Free Radicals: Internal and External Sources
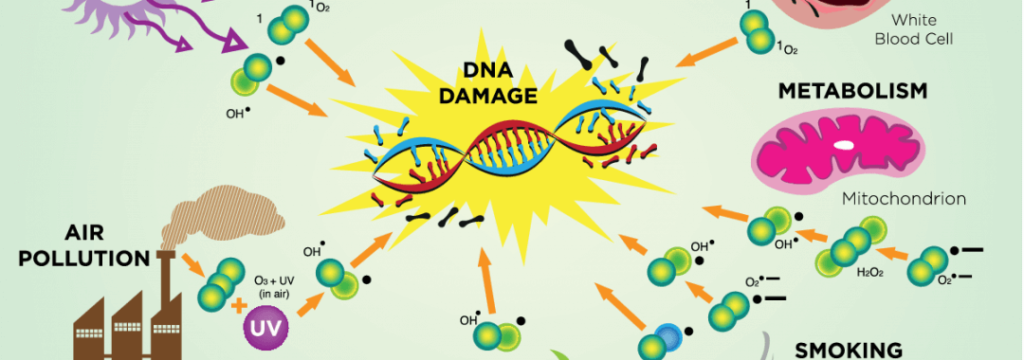
Internal Sources of Free Radicals
| Inflammation: Inflammatory processes within the body can trigger the generation of free radicals. Exercise: Physical activity, while beneficial for overall health, can lead to the internal production of free radicals. Mitochondria: The respiratory chain within mitochondria is a primary internal source of free radicals. Peroxisomes: Peroxisomes, cellular organelles involved in various metabolic processes, contribute to the internal generation of free radicals. Phagocytosis: The immune system’s phagocytic cells generate free radicals as part of their defense mechanisms. |
External Sources of Free Radicals
| Radiation: Exposure to ionizing radiation from sources like sunlight or medical procedures can generate free radicals. Environmental Pollution: Pollutants present in the environment, such as industrial emissions and vehicle exhaust, contribute to the external presence of free radicals. Drugs and Pesticides: Certain pharmaceutical drugs and agricultural pesticides contribute to the external pool of free radicals. Cigarette Smoke: Tobacco smoke, a known source of numerous harmful compounds, including free radicals, poses a significant external threat. Ozone Layer: Even the protective ozone layer, when compromised, can serve as an external source of free radicals, especially at high altitudes. |
Types of Free Radicals
Several distinct types of free radicals are identifiable:
| Superoxide Radicals (O-2): Produced through cellular metabolic reactions, superoxide radicals serve as essential agents in the bactericidal actions of phagocytes—a type of immune cell. However, these radicals can also act as harmful mediators, contributing to inflammation and causing damage to normal body tissues. Hydroxyl Radicals (OH-): Formed during various cellular chemical reactions involving hydrogen, hydroxyl radicals are the most reactive among free radicals. They stand out as major contributors to cell damage and are involved in critical cellular processes. Nitric Oxide (NO): Characterized by a short lifespan, nitric oxide is a highly diffusible, lipid-soluble radical. Despite its brief existence, NO plays a crucial role in promoting immune defense, establishing itself as a significant free radical within the body. |
Classifying Free Radicals
Primary Free Radicals: Generated through the transfer of electrons to oxygen atoms, these radicals showcase an exceptionally brief average lifespan. Originating from enzymatic systems, they fulfill essential roles in various biologically significant functions.
Secondary Free Radicals: Originating from the transfer of a primary radical to an atom of an organic molecule or the interaction of two primary radicals, secondary free radicals boast an extended average lifespan compared to their primary counterparts. Their formation is triggered by reactions between divalent iron ions and hydroperoxides, resulting in the damage of cell structures.
Stable Free-Radical Intermediates: Distinct from traditional radicals, stable free-radical intermediates represent molecules that are not radicals themselves. However, they serve as precursors or sources from which free radicals can emerge. While these intermediates possess stability, their role in facilitating radical formation adds a nuanced layer to the intricacies of free radical chemistry.
Aging’s Silent Aggressor: The Devastation of Free Radicals
The mid-20th century witnessed a revolutionary concept: the free radical theory of aging. This theory linked the destructive power of free radicals to cellular breakdown, offering a new lens on aging. It proposes that as we age, our ability to neutralize these free radicals weakens. This imbalance leads to “oxidative stress,” a cellular assault that fuels age-related decline. Understanding this process is key to unlocking the secrets of aging.
- Free Radicals and Aging: Free radicals, generated by the body’s natural processes, can damage cells.
- Oxidative Stress: When free radical production overwhelms the body’s antioxidant defenses, oxidative stress occurs.
- Cellular Damage: Oxidative stress damages vital cellular components like proteins, lipids, and DNA.
- Impact on Health: This damage contributes to various age-related diseases like cancer, diabetes, and neurodegenerative disorders.
- Importance of Management: Understanding and managing oxidative stress is crucial for promoting overall health and potentially mitigating age-related risks.

Oxidative Damage to Protein and DNA
Protein Oxidation
Proteins are vulnerable to oxidative modifications through processes such as specific amino acid oxidation, free radical-mediated peptide cleavage, and cross-linkage due to lipid peroxidation products. Methionine, cysteine, arginine, and histidine amino acids are particularly susceptible. Oxidative damage to proteins affects enzyme activity, membrane transport, and contributes to aging. Peroxyl radicals and reactive oxygen species (ROS) play a key role in protein oxidation, producing compounds like carbonyls and methionine sulfoxide.
DNA Damage
DNA and RNA are susceptible to oxidative damage, especially in aging and cancer. The levels of oxidative nucleotides rise, contributing to DNA damage, particularly the modified DNA nucleotide 8-hydroxy-2-deoxyguanosine. Mitochondrial DNA is particularly sensitive to oxidative damage, linking it to various diseases, including cancer. This modified DNA nucleotide serves as a biomarker for oxidative stress, offering insights into conditions like aging and cancer. The intersection of oxidative stress with cellular components underscores its significant role in influencing aging and disease development.
Lipid Peroxidation
Oxidative stress and biomolecule modification are involved in physiological and pathophysiological processes, including aging, inflammation, and carcinogenesis. Lipid peroxidation, a free radical process, occurs on cell membranes’ polyunsaturated fatty acids, generating compounds like malondialdehyde. These compounds serve as markers in lipid peroxidation assays and are linked to diseases such as neurodegenerative disorders and diabetes.
oxidative stress and neurological disorders
Oxidative stress, a consequence of free radical overproduction, significantly impacts neurons. These free radicals, generated during cellular processes, unleash a wave of cellular damage. They target vital components like cell membranes, DNA, and proteins, potentially leading to neuronal death.
This damage is linked to several neurological disorders:
- Alzheimer’s Disease: Oxidative stress fuels the formation of protein deposits that disrupt memory and thinking. Targeting this process may offer avenues to slow disease progression.
- Parkinson’s Disease: The degeneration of dopamine-producing neurons, a hallmark of Parkinson’s, is linked to oxidative stress. Understanding and mitigating this stress could lead to potential interventions.
- Amyotrophic Lateral Sclerosis (ALS): Oxidative stress contributes to the progressive loss of motor neurons in ALS, leading to muscle weakness. Researching its role in ALS pathogenesis may unlock novel therapeutic strategies.
Foods Fortifying Against Free Radicals
The good news is, your body doesn’t fight free radicals alone. A diverse range of foods acts as powerful allies. Berries, packed with antioxidants, neutralize these harmful molecules. Leafy greens offer compounds that combat oxidative stress. Nuts, seeds, and vitamin C-rich fruits further bolster your body’s defenses. By incorporating these antioxidant powerhouses, you can fortify your cells against free radical damage, promoting overall health and vitality.
Frequently Asked Questions
Q1: What are free radicals, and how are they formed?
A1: Free radicals are highly reactive molecules characterized by unpaired electrons. They form when molecules undergo a process where they split into smaller particles, leaving one or more electrons unpaired. Common sources include exposure to UV radiation, pollution, and certain metabolic processes within the body.
Q2: Why are free radicals considered harmful to the body?
A2: Free radicals are harmful because their unpaired electrons make them chemically unstable. In an attempt to stabilize, they react with other molecules in the body, leading to oxidative stress. This chain reaction of damage is associated with various diseases, including cancer, cardiovascular disorders, and premature aging.
Q3: How does the body naturally defend against free radicals?
A3: The body employs antioxidants as a natural defense against free radicals. Antioxidants neutralize these molecules by donating electrons without becoming destabilized themselves. Examples of crucial antioxidants include vitamins C and E, along with enzymes like superoxide dismutase, which collectively help maintain balance and protect against oxidative stress.
Q4: What role do free radicals play in aging, and can their impact be minimized?
A4: Free radicals contribute to the aging process by causing cellular damage over time. To minimize their impact, adopting a healthy lifestyle is crucial. This includes a balanced diet rich in antioxidants, engaging in regular exercise, and avoiding excessive exposure to environmental factors that generate free radicals, such as tobacco smoke and UV radiation. These measures support the body’s natural defenses, promoting overall well-being.







